-
Products
-
- Featured Products
- Supraflex Cruz
-
- Professionals
- Patients & Caregivers
- Investors
- About SMT
Main navigation
Platinum nanocoating technology
Nitinol wire used in the Cocoon Duct Occluder is coated with Platinum atoms using nano fusion technology by plasma deposition
Raw nitinol

Electropolished

Platinum nanocoating

Platinum nanocoating makes the Cocoon Duct Occluder:
Biocompatible
Non-corrosive
Non-allergic
More radiopaque
Key highlights
- Hat-shaped design ensures secured implantation at the aorta and minimizes risk of migration into pulmonary artery
- Unique Platinum nanocoating
- Device is filled with polypropylene fabric to assist thrombogenicity
- Extended retention portion provides proper positioning of the device in the ductus arteriosus
- 100% occlusion rate at one-month follow-up1
- Ability to recapture and redeploy*
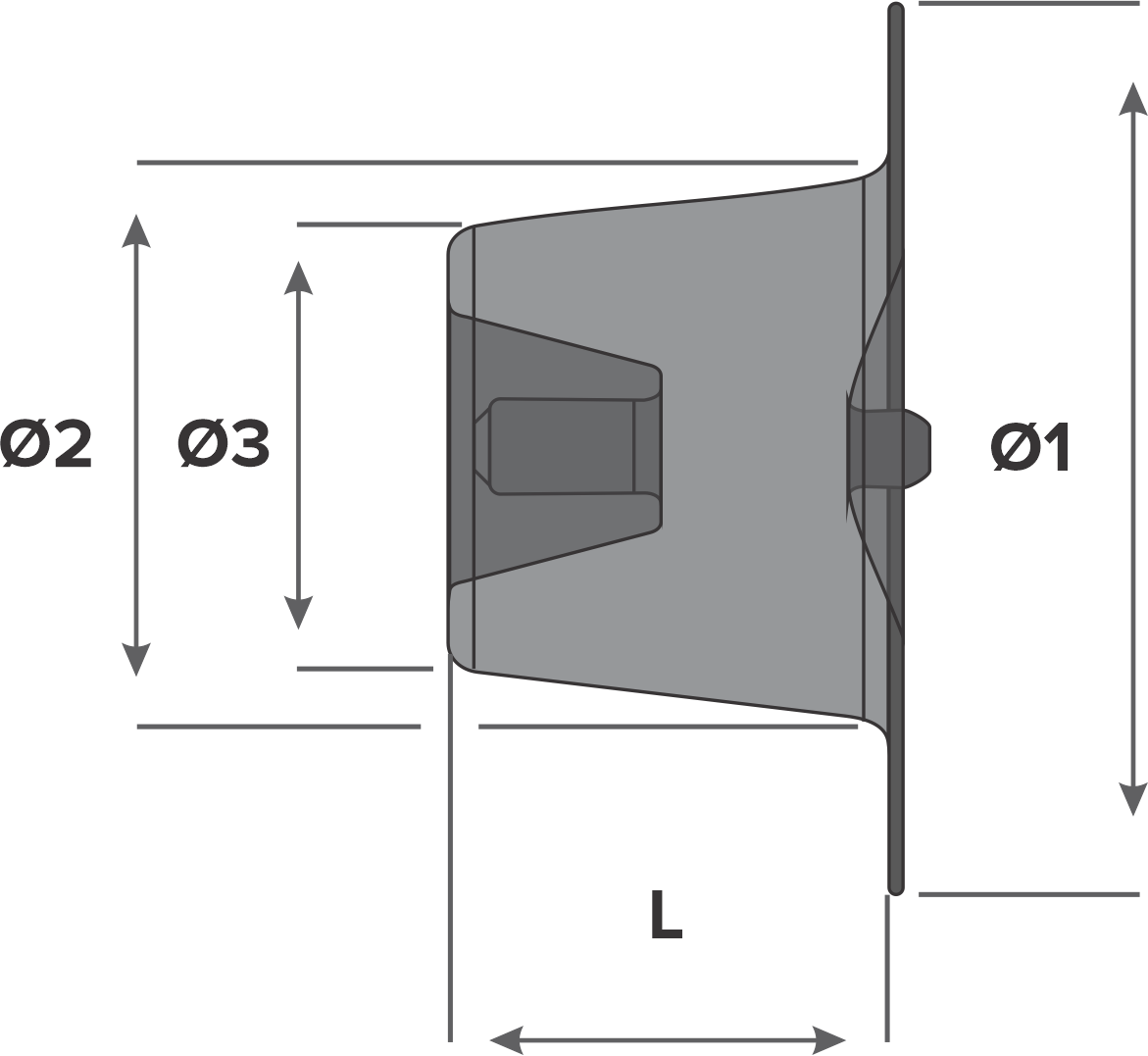
Design:
- Retention skirt diameter (Ø1)
- Device diameter at descending aorta (Ø2)
- Device diameter at pulmonary artery (Ø3)
- Device length (L)
*Recapture and redeployment is possible only if delivery cable is securely connected to the disc.
Technical specifications
| Product Code | Retention skirt diameter (Ø1) (mm) |
Device diameter at descending aorta (Ø2) (mm) |
Device diameter at pulmonary artery (Ø3) (mm) |
Device length (L) (mm) |
CompatibleSheath Size (F) |
|---|---|---|---|---|---|
| COP0406 | 10 | 6 | 4 | 7 | 6-7 |
| COP0608 | 12 | 8 | 6 | 7 | 6-7 |
| COP0810 | 16 | 10 | 8 | 8 | 7-8 |
| COP1012 | 18 | 12 | 10 | 8 | 7-8 |
| COP1214 | 20 | 14 | 12 | 8 | 8-9 |
| COP1416 | 22 | 16 | 14 | 8 | 8-9 |
| COP1618 | 24 | 18 | 16 | 8 | 9 |
| COP1820 | 26 | 20 | 18 | 8 | 9 |
Usable lengths of compatible accessories (cm)
| Usable Length (cm) | ||||
|---|---|---|---|---|
| 6F | 7F | 8F | 9F | |
| Sheath | 83 | 83 | 83 | 83 |
| Dilator | 89 | 89 | 89 | 89 |
| Loader | 10 | 10 | 10 | 12 |
Delivery cable
| Description | Profile (inch) | Length (cm) | Catalog Number |
|---|---|---|---|
| 5F Delivery Cable | 0.043 | 125 | CDC5F |
| 6F Delivery Cable | 0.077 | 117 | CDC6F |

Preclinical studies
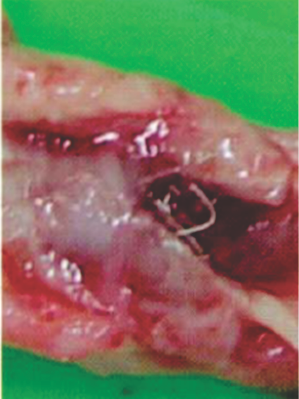
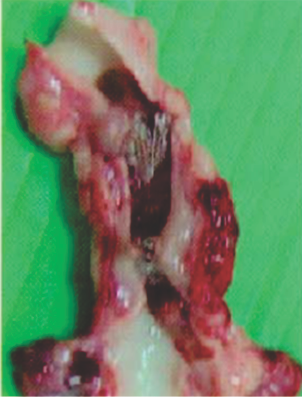
Seven days after implantation in a pig PDA shows a well-encapsulated device
PDA: Patent Ductus Arteriosus
References:
1. Medicine (Baltimore). 2019 Mar; 98(10): e14684.
*Data on SMT file.
#Recapture and redeployment is possible only if delivery cable is securely connected to the disc
Caution: This product is intended for use by or under the direction of a physician. Prior to use, refer to the "Instructions for use" supplied with these devices for indications, contraindications, side effects, suggested procedure warnings and precautions. As part of our continuous product development policy, we reserve the right to change product specifications without prior notification. Information contained herein is for distribution outside the USA & Japan.
Check the regulatory status of the device before distribution in areas where CE marking is not the regulation in force. Tests performed by and data on file at Sahajanand Medical Technologies Limited (SMT). Illustrations are artist’s representations only and should not be considered as engineering drawings or photographs. Photos on file at Sahajanand Medical Technologies Limited.
Cocoon Duct Occluder is manufactured by Vascular Innovations Co., Ltd. Thailand, a SMT group company. Cocoon is a trademark of Vascular Innovations Co., Ltd.
Cocoon range of occluders are currently not approved by USFDA and are not available for sale in USA.
Disclaimer: © 2025 Sahajanand Medical Technologies Limited – All rights reserved. Specifications are subject to modification, revision and improvement.


